Article Text
Abstract
Colorectal cancer (CRC) is the second leading cause of cancer death in USA, and CRC screening remains suboptimal. The aim of this quality improvement was to increase CRC screening in the internal medicine clinic (IMC) patients, between the ages of 50–75 years, from a baseline rate of 50%–70% over 12 months with the introduction of faecal immunochemical test (FIT) testing. We used the Plan–Do–Study–Act (PDSA) method and performed a root cause analysis to identify barriers to acceptance of CRC screening. The quality improvement team created a driver diagram to identify and prioritise change ideas. We developed a process flow map to optimise opportunities to improve CRC screening. We performed eight PDSA cycles. The major components of interventions included: (1) leveraging health information technology; (2) optimising team work, (3) education to patient, physicians and IMC staff, (4) use of patient navigator for tracking FIT completion and (5) interactive workshops for the staff and physicians to learn motivational interview techniques. The outcome measure included CRC screening rates with either FIT or colonoscopy. The process measures included FIT order and completion rates. Data were analysed using a statistical process control and run charts. Four hundred and seven patients visiting the IMC were offered FIT, and 252 (62%) completed the test. Twenty-two (8.7%) of patients were FIT positive, 14 of those (63.6%) underwent a subsequent diagnostic colonoscopy. We achieved 75% CRC screening with FIT or colonoscopy within 12 months and exceeded our goal. Successful strategies included engaging the leadership, the front-line staff and a highly effective multidisciplinary team. For average-risk patients, FIT was the preferred method of screening. We were able to sustain a CRC screening rate of 75% during the 6-month postproject period. Sustainable annual FIT is required for successful CRC screening.
- primary care
- quality improvement
- root cause analysis
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.
Statistics from Altmetric.com
Introduction
An estimated 137 000 new cases of colorectal cancer (CRC) and 50 260 related deaths occurred in 2017.1 CRC screening significantly reduces the incidence and mortality of this disease2–5; however, it remains suboptimal, particularly among the underserved population.6–8 In the academic, safety-net Internal Medicine Clinic (IMC) at Erie County Medical Center (ECMC), less than 50% of active, eligible patients were screened for CRC by December of 2016. In March 2014, the American Cancer Society (ACS), the Centers for Disease Control and Prevention and the National Colorectal Cancer Roundtable (NCCRT) proposed The ‘80% by 2018’ initiative with a goal of implementing CRC screening for 80% of adults between the ages of 50 years and 75 years by 2018.9 10 The ECMC leadership pledged 80% CRC screening by 2018; therefore, we designed this quality improvement (QI) to increase the CRC screening in the IMC population.
Preliminary studies suggest that biennial screening instead of annual screening may be effective.11 12 However, at this time, the US Preventive Service Task Force screening recommendations include colonoscopy (every 10 years) or home-based faecal testing (every year) for average-risk adults.13–17 Many individuals are asymptomatic early in the disease course due to the slow growth of precancerous polyps to invasive cancer. Screening allows for the opportunity for early detection, removal of precancerous polyps and prevention of CRC.3 4 18 19 The faecal immunochemical test (FIT) is a less expensive, non-invasive alternative to colonoscopy,20 21 which uses antibodies specific for human haemoglobin to reveal haemoglobin in faecal occult blood.22–25 Currently, FIT is the most commonly used method for CRC screening in average-risk patients26 27; greater adherence to this test is due to fewer stool samplings and lack of dietary or medication restrictions.22 24 28 29 The aim of this QI was to increase CRC screening in the IMC from the baseline rate of 50%–70% in patients between the ages of 50 years and 75 years over 12 months with the introduction of FIT.
Methods
Setting
We conducted a QI project in an academic IMC, located within a tertiary care safety-net hospital, ECMC. The IMC patient population consists of mostly urban, underprivileged and African-Americans (68.42%). Patients use the IMC as a longitudinal primary care clinic; the IMC has an average of 700 monthly visits. The IMC is composed of a multidisciplinary care team including 35 residents from the University at Buffalo’s Internal Medicine Residency Program and four attending physicians.
Design
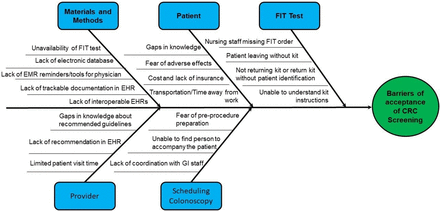
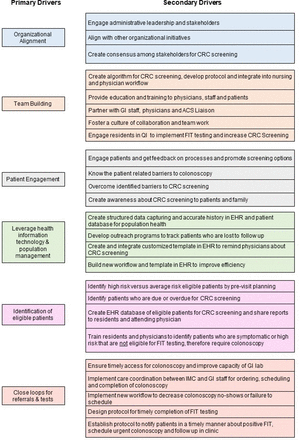
We designed this QI based on the Plan–Do–Study–Act (PDSA) model of healthcare improvement.30 31 The QI team included a physician champion, nursing and ancillary staff, residents, attending physicians, a social worker, gastroenterolgy (GI) nurse practitioner (NP), a patient navigator, patients, administrative and IT staff and a project liaison from the ACS. The QI team performed a root cause analysis using a fishbone diagram and identified the materials/methods, physician and patient-related barriers to the acceptance of FIT and colonoscopy (figure 1). The QI team identified primary and secondary drivers, brainstormed about potential change ideas and created a driver diagram in order to accomplish our aim32 33 (figure 2). We identified strengths and prioritised change ideas to overcome the challenges to improve screening rates (table 1). We developed a new process flow map to optimise opportunity to improve CRC screening (figure 3).
Fishbone diaphragm: root cause analysis identifying barriers to acceptance of CRC screening. CRC, colorectal cancer; FIT, faecal immunochemical test; EHR, electronic health record; EMR, electronic medical record.
CRC screening driver diagram. ACS, American Cancer Society; CRC, colorectal cancer; FIT, faecal immunochemical test; IMC, internal medicine clinic; QI, quality improvement; GI, gastroenterology; EHR, electronic health record.
Process flow map. FIT, faecal immunochemical test; CRC-2, colorectal cancer screen 2
Change ideas tested by IMC
Inclusion criteria consisted of asymptomatic male and female patients, between the ages of 50 years and 75 years for CRC screening with (A) no prior colonoscopy, (B) had a colonoscopy greater than 10 years ago and due for a rescreen and (C) had a colonoscopy less than 10 years ago with a diagnosis of precancerous polyp; therefore, patient was due for a rescreen. Patients were excluded if they had a colonoscopy with normal findings within 10 years or had a negative FIT within 1 year. In December 2016, the ECMC laboratory implemented a hema-screen FIT. The sensitivity cut-off is 50 µg Hb/g of faeces.34 The sensitivity, specificity, positive likelihood ratio and negative likelihood ratio of a single FIT for cancer has been shown to 0.79, 0.94, 13.10 and 0.23 µ g Hb/g of faeces, respectively.35 Physicians discussed the FIT-positive results with the patients and referred the patients for subsequent diagnostic colonoscopy. The physician scheduled a clinic visit to evaluate the barriers to acceptance of colonoscopy after patients missed scheduled colonoscopy appointment. Our CRC screening rates for FIT or colonoscopy were based on the review of completed test reports by the physician.
Measurements
We created an electronic patient database in collaboration with the information technology (IT) department. The baseline CRC screening rate was 50% in December 2016, obtained by retrospective review of the database of active, eligible patients seen at least once within the past 18 months in IMC. The outcome measures included: (1) increase in CRC screening rates to 70% with either FIT or colonoscopy from the baseline rate and (2) diagnostic colonoscopy completion rate in FIT-positive patients. The process measures included FIT order and completion rates. The balance measures were an increase in patient wait time during the IMC visit and poor access for timely screening colonoscopy. The majority of average risk patients refused colonoscopy and preferred the FIT. Therefore, we selected FIT order and completion rates as process measures. CRC screening by FIT is only effective in identifying early CRC or preventing CRC by timely diagnostic colonoscopy in FIT-positive patients.17 19 Therefore, we included subsequent colonoscopy in positive FIT patients for our outcome measures. The QI team identified multiple challenges for colonoscopy including lack of patient’s preference and a long wait time due to limited capacity from increased demand for CRC screening for high-risk patients.
Strategy
PDSA cycle 1 (December 2016–January 2017): customised template in the EHR and physician training
We created a customised EHR template that included a FIT check list for the physician. This list included: (1) FIT or colonoscopy option; (2) discussion with patient about the need for colonoscopy after a positive FIT; and (3) discussion with patient about annual FIT after a negative FIT. We identified gap in resident’s knowledge of CRC screening. We educated physicians and clinic staff through PowerPoint presentations in a small group discussion. This curriculum included current CRC guidelines and EHR workflow integration for FIT.
PDSA cycle 2 (February 2017): physician and staff reminder, FIT instruction and patient navigator
Physician and staff reminder
There was a lack of automated EMR chart alerts for the physician to identify patients that needed CRC screening. We implemented a new workflow that consisted of completion of the CRC-2 questionnaire by the patients with facilitation of the nursing staff prior to the physician evaluation. The CRC-2 questionnaires included the following questions: (1) Have you ever had a colonoscopy?, (2) Are you willing to discuss CRC screening with your physician today? This workflow was designed to identify patients and to remind the physicians to discuss the CRC screening. Initially, patients completed CRC-2 questionnaire during registration and the nursing staff entered this information into the EHR. After a few weeks, the nursing staff administered and entered the CRC-2 questionnaire into the EHR and also entered a chart alert for the physicians. The QI team developed a ‘FIT’ stamper, and physicians were instructed to stamp the billing discharge paper to remind the nursing staff to dispense the FIT KIT prior to the patient being discharged.
FIT instruction
Most of the patients left without a FIT kit due to inability to watch the 5 min video regarding CRC screening and FIT instruction. We replaced the video with a simplified one-page paper instruction that described the three steps of the FIT and also included a reminder to return FIT kit within 3 days.
Patient reminders by navigator
A patient navigator tracked FIT orders in the EHR database and contacted patients weekly with a reminder phone call if they did not return FIT kits.
PDSA cycle 3 (March 2017): interactive workshop
The physician champion conducted an interactive workshop36–38 using the NCCRT handbook,39 engaging the IMC multidisciplinary team. A pretest and post-test was administered to identify gaps in the physician’s knowledge and to evaluate improvement. The pretest consisted of five questions about CRC statistics, barriers, target audiences and top-rated clinical messages to screening. Professional role play scenarios were created by the IMC staff; and residents were assigned to motivate patient to make a shared patient-centred decision for CRC screening.
PDSA cycle 4 (April–May 2017): EHR outage
On 9 April 2017, ECMC faced an EHR outage for 2 months. FIT was not available for 1 month. The QI team continued to share progress of this QI with IMC staff.
PDSA cycle 5 (June 2017): patient education and tracking of FIT orders
Patient education materials were placed in the examination rooms to create awareness about CRC screening. The QI team outlined the IMC protocol for tracking of FIT orders by the patient navigator. The patient navigator contacted the patient weekly for three attempts, followed by a letter to remind the patient to return the FIT kit. The patient navigator cancelled the FIT order after no response and notified the physician about FIT order status. The patient navigator documented this process in the patient’s EHR.
PDSA cycle 6 (July–August 2017): physician education and patient reminder
Initial and refresher training for the residents was conducted for CRC screening. The patient navigator continued to track FIT orders and remind patients to return FIT kits.
PDSA cycle 7 (September–October 2017): FIT incentive and tracking FIT orders
We introduced a FIT incentive programme ($5 gift card) for timely completion of the FIT kits.
PDSA cycle 8 (November–December 2017): interactive workshop, patient education and physician reminder
The interactive workshop was conducted for the residents. We created a pocket card for physicians that included a CRC screening algorithm and the process flow map for CRC screening. Patient education pamphlets were placed in the examination room to create CRC awareness.
Data analysis
Data were analysed using statistical process control charts for process measures and run chart for outcome measure of CRC Screening. A paired samples t-test was used to compare the mean percentage of correct answers between pretest and post-test.
Results
Demographics
A total of 407 patients received FIT kits. This population had a mean age of 61.3, was 49.4% female, 64.9% were African-Americans, 30.7% were white while 4.4% were classified as other race. We performed a demographic comparison of patients that completed the FIT (group 1) versus those that did not (group 2); 252 completed the test, while 155 did not complete the test. Those in group 1 were 51.2% female, with a mean age of 61.5, 65.5% were African-American, 29.4% were white and 5.2% were classified as other race. Furthermore, in group 1, 17.1% of patients had a history of serious mental illness (schizophrenia or bipolar disorder). For group 2, patients were 46.4% female, with a mean age of 60.9, 63.9% were African-American, 32.9% were white and 32% were classified as other race. Additionally, in group 2, 11.0% of patients had a history of serious mental illness. We did not observe any significant difference in age, gender, race and history of serious mental illness in patients who completed FITs.
Process measures
FIT order rate
The mean FIT order rate was 25.6% (figure 4A). We observed significant weekly variations in FIT order rates during various PDSA cycles and a sustainable increase in specific weeks within 12 months.
(A) Weekly statistical process control (SPC) chart showing percentage of FIT order rate. (B) Monthly SPC chart showing percentage of FIT completion rate. CL, control limit; FIT, faecal immunochemical test; LCL, lower control limit; PDSA, Plan–Do–Study–Act; UCL, upper control limit.
FIT completion rate
The mean FIT monthly completion rate was 66.2% (figure 4B). The mean time from FIT dispense to results was 27.1±37.0 days and median time from FIT dispense to results was 14.0±23.0 days (IQR). Sixty-two per cent (n=252/407) of patients completed FIT. Out of these patients, about 68% (n=172) were screened for initial CRC and 32% (n=80) were screened for repeat CRC. Financial incentives did not significantly improve the FIT completion rate.
Improvement in physician knowledge after interactive workshop
We conducted a pretest and post-test to assess objective evidence of improvement in residents’ knowledge. Results showed a statistically significant improvement in the mean percentage of correct responses between pretest and post-test answers (n=32). The mean percentage of correct pretest answers was 48.1%±25.8, which improved to 96.3%±7.9 (p<0.001) post-test.
Outcome measures
CRC combined (FIT and colonoscopy) screening rate
The median CRC combined rate (with either FIT or colonoscopy) in the run chart was 74.6% from December 2016 to December 2017 (figure 5). We achieved a sustainable increase in the CRC screening rate to 74.94% (3428/4574 visits), with either FIT or colonoscopy within 12 months (January–December 2017). Positive FIT patients who did not get subsequent diagnostic colonoscopy were not counted for CRC screening rates, since their screening status remained incomplete. We achieved 70.6% (n=1238/1754) CRC screening by FIT or colonoscopy in the active (seen within past 18 months) IMC patient population by December 2017. We are able to sustain a CRC screening rate of 75% during the 6-month postproject period (January–July 2018).
Monthly CRC combined Screening Run chart 2017. CRC, colorectal cancer; PDSA, Plan–Do–Study–Act.
Colonoscopy completion rate
There was an increase in referrals for screening colonoscopy for high-risk patients during 12 months. The colonoscopy completion rate was 64% (n=228/358) from January 2107 to December 2017. The colonoscopy completion rate also included diagnostic colonoscopy. However, the majority of patients for diagnostic colonoscopy did not have any prior CRC screening.
FIT-positive rate
The FIT-positive rate was 8.7% (n=22/252) and 14 of those (63.6%) underwent subsequent diagnostic colonoscopy. All the patients completed colonoscopy within 3 months, except two patient completed within 8 months. One patient was not appropriate for a repeat colonoscopy due to recent colonoscopy in 2015, and one patient has pending colonoscopy. Colonoscopy appointments were rescheduled in six patients after the IMC physicians re-evaluated and discussed the need for diagnostic colonoscopy with patients. Patients had agreed for the colonoscopy; however, they did not keep scheduled colonoscopy appointments. The majority of the patients (4/6) had serious mental illness (schizophrenia or bipolar disorder), and they declined colonoscopy due to the fear of the procedure. Two patients had agreed for an alternate radiology test (double contrast barium CT scan), and one completed the test, but it was a poor study due to large amount of stool present and the other patient did not keep the scheduled appointment. In the FIT-positive IMC population, we found no active cancer, 50% average risk patients had precancerous polyps. Diagnostic colonoscopy results showed tubular adenoma in six patients, tubulovillous adenoma in one patient, diverticulosis in three patients, normal findings in two patients and normal colon with haemorrhoids in one patient. One patient needed a repeat colonoscopy due to poor preparation. One hundrd and twenty-five FITs were completed, and 8.0% (10/125) were positive FIT during the 6 month postproject period. Fifty per cent (5/10) of these FIT-positive patients had subsequent colonoscopy, and the other patients are scheduled for colonoscopy.
Balance measures
Increase in wait time for patient
The extra time used on the screening tool did not result in any increase in patient wait time in the clinic. This was determined by anecdotal reports of lack of patient’s complaints for a long wait time and lack of overtime for clinic staff during this project. The average time spent in the clinic during a follow-up visit was about 1 hour prior to this QI. The time did not change during project. We did not measure actual time wait time during this QI project.
Increase in wait time for screening colonoscopy
Due to an increase in demand for screening colonoscopy and lack of sufficient GI physicians, there was an increase in the wait time. Before this QI, the average wait time for a screening colonoscopy at ECMC was about 3 months, and it doubled initially after the implementation of this QI due to increase in colonoscopy referrals.
Discussion
We identified a gap in CRC screening at safety-net IMC at ECMC. This QI project was implemented in response to the NCCRT ‘80% by 2018’ CRC screening initiative.10 The aim of this QI was to increase CRC screening from the baseline rate of 50%–70% in the IMC population over the period of 12 months with the introduction of the FIT. We were able to demonstrate meaningful impact from the different PDSA cycles during this QI. We achieved a sustainable increase in CRC screening rate with either FIT or colonoscopy to 75% and surpassed our goal of 70% within a short period of 12 months (January–December 2017). In the IMC, 8.7% of the patients screened for CRC had positive FITs. Of those FIT-positive patients, 63.6% completed colonoscopy within 3–6 months. Studies have shown that the time to colonoscopy can result in a negative impact on the patient’s health.40 41 Delays of up to 12 months after a FIT-positive result can produce up to nearly 10% in losses in overall screening benefits.40
The success of this QI project was attributed to patient education, interactive workshops and patient outreach by a navigator. There was objective evidence of increased physician knowledge related to CRC screening after implementation of interactive workshops. Several other initiatives have had relatively positive success rates in improving CRC screening using FIT,42–46 patient5 47 48 and physician education.49–51 Patient outreach by a navigator had a positive impact on FIT completion rates similar to previous studies.46 52–54 A decline in the FIT completion rate in the last few months in 2017 may be attributed to the lack of sufficient time allocated to the patient navigator due to shortage of staffing in the IMC.
Patients were actively engaged in this QI project in identification of barriers to CRC screening, education, shared decision making for CRC screening options and feedback on various change ideas. Physicians understood patients’ related challenges to CRC screening by embracing open communication with the patients. A social worker interacted with the patient during the clinic visit and offered solutions to overcome barriers. Additionally, a patient navigator assessed the patient-related barriers for FIT completion by calling patients. We identified two major barriers to the completion of FIT: lack of interest due to knowledge gaps and forgetting to do the test. The nursing staff and physicians educated patients on CRC and screening options and facilitated a shared decision to determine the method of CRC screening. After receiving feedback from the patients, the QI team simplified FIT instructions and offered a patient incentive for FIT completion. We were able to update family history of CRC and retrieve reports of prior colonoscopy after physician’s interaction with patients during clinic visit. The team implemented a new workflow of precolonoscopy consultation with a GI NP to overcome the barrier of fear that helped to improve patient’s understanding and adherence to colonoscopy.
Lessons learnt
Critical factors for success
We attributed the success of this QI to the three T’s: (1) technology, (2) tactics and (3) team. Engagement of a multidisciplinary team in performing the root cause analysis and creating the driver diagram was crucial to the design of this QI.
Learning from failures
We offered a $5 financial incentive, which did not increase our FIT completion rates. Financial incentives lower than $50 has been shown not to increase FIT completion rates.55–57 Established barriers to colonoscopy such as transportation and financial considerations were not major barriers in this QI.19 58 Viewing of videos on FIT instructions by patients was not successful in helping the patient to understand the procedure; this led to the creation of simplified paper instructions, designed with patient’s feedback. Initially, the nurses were missing FIT orders in the EHR; therefore, the team created a visual reminder using a red colour ‘FIT’ stamper on the discharge paper for the nursing staff. We were unable to reach patients for notification of positive FIT results due to inaccurate phone numbers, so we reminded staff to update the patients’ contact information during clinic visit. Diagnostic colonoscopy for FIT-positive patients were not scheduled in a timely manner by placing urgent referral in EHR, therefore in addition to a referral, the team designed a new workflow where the physician sent a task directly to the GI NP to facilitate urgent colonoscopy. Initially, we observed multiple no-shows for scheduled colonoscopy, so the team implemented a precolonoscopy consultation appointment with the GI NP to review bowel preparation and procedure with patients. The team improved communication between the GI staff and IMC physicians by adding a new process of notification of no-shows of colonoscopy to the physicians by sending task in the EHR. Due to an increase in demand for colonoscopy, the ECMC leadership added two additional physicians to assist in colonoscopy to improve timely access. During this QI, we discovered inaccuracies in the database for CRC screening, through collaboration with a highly engaged IT staff we were able to ensure accuracy of the database.
Challenges
A few team members proposed using an iPad in the examination room for patient education to show videos and PowerPoint slides for messages for CRC screening. However, due to the cost and safety of iPad in the clinic, the team decided to purchase digital photo frames. Digital photo frames worked well; patients were engaged in watching CRC educational material in the examination room while waiting for the physician. Open and honest communication among multidisciplinary team members facilitated challenging discussions, and the team agreed to try new changes as a pilot test even after there was disagreement among members. The team learnt about various new ideas and embraced successes and failures. The team meetings with IMC residents, physicians and interdisciplinary team, held every 5 weeks, enhanced communication and learning that led to redesigning different interventions.
Previsit planning by the nursing staff to identify patients due for CRC screening was not successful due to time constraints and limited resources. The team agreed to change this workflow and instructed physicians to review and retrieve medical records of prior colonoscopy. Furthermore, we implemented a medical office assistant (MOA) workflow whereby the MOA entered colonoscopy and FIT reports, refusal for both tests and a reminder for a repeat colonoscopy as a structured data field in the EHR to improve accuracy of EHR database. However, due to limited resources, this workflow was not sustainable. Physicians were trained to enter this information in the EHR, and due to physician’s time constrains, it was not entered in the majority of patients. The team planned to reach out to patients who were lost to follow-up for population health, but due to lack of resources, we were unable to accomplish patient outreach. The lack of clinical decision support tool for the physician to identify patients and order appropriate CRC screening test was found to be the greatest IT challenge. Multiple attempts were made by physicians to review a need for subsequent colonoscopy in FIT-positive patients; however, physicians identified major barrier of serious mental illness (schizophrenia and bipolar disorder) to the acceptance to colonoscopy.
This QI has evidence of internal validity. The average time spent by the patient navigator was about 4 hours per week. There was minimal cost involved in conducting this QI. This QI project has several limitations. We reported the data for this QI at 12 months, and it was difficult to have significant improvements in CRC screening rates using colonoscopy due to a long waiting period for the test. This QI was performed in a safety-net primary care clinic in patients with multiple comorbidities, so the barriers and interventions identified in this QI may not be generalisable to other settings.
Sustainability and spread
Processes and workflows designed during this QI project for improving CRC screening in the IMC have become the standard of care and routine part of the clinic visit. IMC physicians continue to offer FIT or colonoscopy options to average risk patients and colonoscopy to high-risk patients. ECMC leadership developed a culture supportive of QI and innovation and assisted in improving access for screening colonoscopy. Continuous training, education and feedback to IMC physicians, residents and staff have fostered team work. We were able to sustain a CRC screening rate of 75% during the 6-month postproject period (January–July 2018). This QI was expanded to new providers (NPs, physician assistants and physicians) in the IMC hired in 2017–2018. The option for FIT for CRC screening is available to patients in the family medicine clinic at ECMC; however, due to physician time constraints, it is not optimal. The team plans to share QI tools, strategies, successes and challenges learnt in this QI project with the family medicine clinic at ECMC and at academic primary care clinic sites at other hospitals. The physician champion presented seminars on this QI project at the University at Buffalo and shared team experience with the residents and faculty from various hospitals. Strategies that were implemented in this QI can be replicated in other settings to improve CRC screening.
Annual FIT screening is feasible for CRC screening in a safety-net clinic. For average-risk patients, FIT was the preferred method of CRC screening in the IMC. We exceeded our goal, achieving 75% CRC screening with either FIT or colonoscopy within a short period of 12 months. NCCRT’s goal of 80% by 2018 is attainable in the IMC patient population. Successful strategies included engaging leadership, the front-line staff, a highly effective multidisciplinary team and leveraging health information technology. We were able to sustain CRC screening rate of 75% during the 6-month postproject period. Longitudinal adherence with annual FIT is critical to reduce CRC incidence and for early diagnosis of CRC. Future directions will include strategies to promote repeat annual FIT and outreach of patients that were lost to follow-up in the IMC for population health CRC screening.
Acknowledgments
We would like to thank Gregory D. Gudleski, PhD, and staff from the Internal Medicine Clinic, Information Technology and the Gastroenterology clinic.
References
Footnotes
Contributors SYB: study oversight, study concept and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision of the manuscript for important intellectual content and finalisation of manuscript. GA and NN: acquisition of data, analysis and interpretation of data. AM: acquisition of data. JR: interpretation of data, drafting of manuscript, critical revision of the manuscript for important intellectual content and finalisation of manuscript.
Funding ACS for the CHANGE grant to increase CRC screening (SB). Research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under award Number UL1TR001412.
Disclaimer The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Competing interests None declared.
Ethics approval This study was approved by the Human Subjects Institutional Review Board (HSIRB) of the University at Buffalo and was exempt from patient consent. The work was deemed a quality improvement project and not a study on human subjects.
Provenance and peer review Not commissioned; externally peer reviewed.
Data sharing statement An electronic database was setup; contact authors for access.