Article Text
Abstract
A significant incidence of post-procedural deep vein thrombosis (DVT) and pulmonary embolus (PE) was identified in patients undergoing surgery at our hospital. Investigation showed an unreliable peri-operative process leading to patients receiving incorrect or missed venous thromboembolism (VTE) prophylaxis. The Trust had previously participated in a project funded by the Health Foundation using the “Safer Clinical Systems” methodology to assess, diagnose, appraise options, and implement interventions to improve a high risk medication pathway. We applied the methodology from that study to this cohort of patients demonstrating that the same approach could be applied in a different context.
Interventions were linked to the greatest hazards and risks identified during the diagnostic phase. This showed that many surgical elective patients had no VTE risk assessment completed pre-operatively, leading to missed or delayed doses of VTE prophylaxis post-operatively. Collaborative work with stakeholders led to the development of a new process to ensure completion of the VTE risk assessment prior to surgery, which was implemented using the Model for Improvement methodology. The process was supported by the inclusion of a VTE check in the Sign Out element of the WHO Surgical Safety Checklist at the end of surgery, which also ensured that appropriate prophylaxis was prescribed. A standardised operation note including the post-operative VTE plan will be implemented in the near future. At the end of the project VTE risk assessments were completed for 100% of elective surgical patients on admission, compared with 40% in the baseline data.
Baseline data also revealed that processes for chemical and mechanical prophylaxis were not reliable. Hospital wide interventions included standardisation of mechanical prophylaxis devices and anti-thromboembolic stockings (resulting in a cost saving of £52,000), and a Trust wide awareness and education programme. The education included increased emphasis on use of mechanical prophylaxis when chemical prophylaxis was contraindicated. VTE guidelines were also included in the existing junior Doctor guideline App. and a “CLOTS” anticoagulation webpage was developed and published on the hospital intranet.
The improvement in VTE processes resulted in an 80% reduction in hospital associated thrombosis following surgery from 0.2% in January 2014 to 0.04% in December 2015 and a reduction in the number of all hospital associated VTE from a baseline median of 9 per month as of January 2014 to a median of 1 per month by December 2015.
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-commercial License, which permits use, distribution, and reproduction in any medium, provided the original work is properly cited, the use is non commercial and is otherwise in compliance with the license. See:
Statistics from Altmetric.com
Problem
This project was undertaken in the Royal United Hospitals Bath, NHS Foundation Trust, a 565 bed acute hospital, serving a population of around 400,000 people.
The problem was highlighted in 2013 when the Trust alerted on the ‘Dr Foster’ database as an outlier for venous thromboembolic event occurring within 30 days of surgery or procedure performed in our hospital. Dr Foster is a clinical database used by more than half of English hospitals to provide bench marking and risk adjusted analysis of hospital episode data. Further investigation revealed there were some mitigating reasons for this; patients referred to our tertiary pulmonary medicine service with pulmonary hypertension were coded into this group. After eliminating that group of patients however, the Trust still had a higher than expected incidence of VTE compared with national data.
Review of the case notes revealed that the NICE guidance (CG92 Venous thromboembolism: reducing the risk)1 was not reliably implemented, and that there appeared to be no standardised approach to recording and analysing each incident of VTE.
Problems around reliable prescription of medication for elective surgical patients, including completion of the VTE assessment, arose when junior doctors were removed from the pre-operative assessment unit where medication history and pre-operative prescription had previously been performed. As part of a service configuration to improve efficiency, junior doctors were replaced by nurses who could perform patient screening, but could not prescribe. Therefore, the link between screening and prescription was lost. The consequence of this intervention had not been fully considered at the time. In addition, VTE mechanical prophylaxis was not standardised throughout the Trust and there was variable knowledge about VTE management amongst nursing and medical staff.
Background
VTE is an important cause of death in hospital patients; the risk of developing VTE depends on the condition and/or procedure for which the patient is admitted and on any predisposing risk factors such as age, obesity, and concomitant conditions.1 ,2
Treatment of non fatal symptomatic VTE and related long term morbidities is associated with considerable cost to health services worldwide2 ,3 and the inconsistent use of prophylactic measures for VTE in hospital patients has been widely reported.
A UK survey suggested that 71% of patients assessed to be at medium or high risk of developing deep vein thrombosis did not receive any form of mechanical or pharmacological VTE prophylaxis.4 Since that study, despite increased focus on VTE prevention, evidence suggests that the delivery of VTE prophylaxis remains unreliable.3 ,5 The aim of this project was to ensure 100% of patients undergoing elective surgery were screened for their risk of VTE on admission from a baseline of 40%. The aim was to complete the project within one year.
Baseline measurement
The Trust alerted on the ‘Dr Foster’ database as having a higher than expected incidence of post-operative VTE. Baseline Measures were collected on a daily basis by the Admission Suite staff and showed that risk assessment was only performed in 40% of elective surgical patients prior to surgery following a snapshot audit over 2 days of 50 patients in total. This audit measured the number of VTE risk assessments completed and documented by the surgeon prior to the patient leaving for theatre.
Outcome Measures
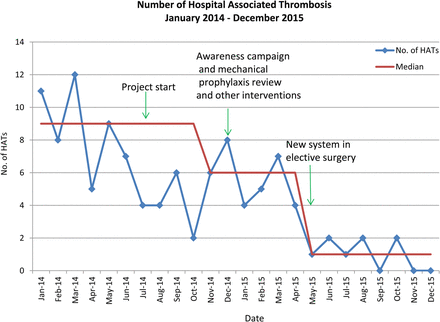
The first outcome measured was the number of Trust wide incidents of Hospital Associated Thrombosis (HAT) in adult patients (figure 1), and the second was the number of Hospital Associated Thrombosis in adult elective surgical patients (figure 2).
Absolute number of all Hospital Associated Thrombosis (HATs): January 2014 (prior to commencing project) to December 2015
Percentage of patients with Hospital Associated Thrombosis per total number of patients undergoing surgical procedure
The Trust had previously implemented an electronic system for recording HATs and undertaking root cause analysis in September 2013. An electronic statement of validation was added in January 2014 to confirm if the VTE incident was hospital associated. Baseline data was available from January 2014.
The healthcare record of all patients with a positive Doppler scan for VTE or CTPA was audited; any patients who were admitted with symptoms of a primary diagnosis of VTE unrelated to surgery were excluded. For those remaining, a full root cause analysis was undertaken and a decision made as to whether the VTE could have been prevented. Cases where the guidelines had not been followed were confirmed as hospital associated thromboses.
As a balancing measure the percentage of returns to theatre due to surgical post-operative haemorrhage were audited. All returns to theatre were reviewed and baseline data showed no obvious incidents of haemorrhage related to thromboprophylaxis.
A small volume of baseline data was collected to ascertain whether patient information in the form of any type of education about the risk of blood clots, prevention and detection had been given to patients and/or carers on admission and following discharge. Patients were surveyed on the pilot wards only. This proved difficult as numbers were too small to analyse and remains a focus for our future work.
Design
Steps one and two of the “Safer Clinical Systems” approach included the set up and diagnostic phases.6 A project team was established supported by funding from the Health Foundation, to improve VTE processes across the Trust. The team comprised of the clinical lead (Haematology Consultant), project nurse (Thrombosis specialist nurse), project lead (Head of Quality Improvement), and project manager (a senior nurse trained in the Safer Clinical Systems approach). Advice and support was sought from the project sponsor (Associate Medical Director for Quality). A specific team was involved in the Elective Surgical Pre assessment pathway which consisted of a Consultant Surgeon, Consultant Anaesthetist, Medical Quality Improvement Lead, and Admission Suite Senior Nurse. Key multi-disciplinary stakeholders from various specialties, including a patient representative, were invited to describe the ‘current state’ and ultimately re-design a new system. Detailed process mapping and diagnostic methodologies such as “Failure, Modes and Effects Analysis”, performance influencing and human factors, and ‘create and detect’ tools were used to understand the hazards and identify the greatest risks in a system.
The results of the diagnostic steps were agreed by the stakeholders and the third phase of the “Safer Clinical Systems” approach - the ‘Options Appraisal’ was used to create a safer system. Solutions and interventions were offered, considered, and scored against specific rationale addressing the reduction of risk as the main priority. Other criteria included ease of implementation, feasibility, sustainability, and financial consequence. The use of this methodology was embraced by the stakeholders as any emotion or bias was removed from the decision making process. The stakeholders needed to agree the scoring and therefore the interventions were more likely to be owned and sustained.
There were two drivers to achieve our aim of reducing hospital associated thrombosis by 30%. These were improvement of the completion of the VTE risk assessment for elective surgical patients to 100%, and to increase the reliable delivery of mechanical and chemical prophylaxis for all patients.
A ‘future state’ hierarchical task analysis was created for the elective surgical pathway detailing the steps to be taken to create a safer system which was shared with the stakeholders and the Hospital Thrombosis Committee. This was then tested using the Plan, Do, Study, Act (PDSA) methodology.
An awareness and educational programme was developed to increase the knowledge and understanding of mechanical and chemical prophylaxis, and increased access to necessary equipment was ensured.
Strategy
The proposed intervention and first PDSA to improve elective surgical VTE risk assessment was for the surgeon to complete the risk assessment in the Admission Suite following patient review. The aim was to test the new process initially in two specialties, with engaged surgeons. Baseline data was collected and the intervention was to feedback this data to all medical staff in the two specialties along with presentation and explanation of the new process. Measures of compliance with VTE risk assessment completion in the two specialties were collected for all patients over one week.
We hypothesised that this would lead to a significant improvement in compliance but results demonstrated no improvement with only 40% of risk assessments being performed.
To understand this fully, feedback was sought from the teams involved. It became clear that many surgeons did not know where the risk assessment was to be found, or how to complete it, despite being informed previously. It was also easy for them to forget as the admission suite is a very busy area.
A second PDSA was tested with several interventions with the aim of supporting completion of the risk assessment.
The intervention was to facilitate the completion of the VTE risk assessment in the Admission Suite where the process was explained to the surgeons by the project manager and any issues resolved in real time, thus supporting reliable completion of the risk assessment. Posters with prompts and instructions to complete the VTE risk assessment were also displayed in the Admission Suite consulting rooms, and the Admission Suite staff also became very engaged and inventive, placing reminder notes on the patient's consent form to prompt VTE risk assessment completion before the consultants left the Admission Suite. This was further supported by the Theatre Orderlies who ensured the VTE risk assessment had been completed before the patients were taken to theatre.
A further intervention occurred at the same time when an additional check at the Sign Out of the WHO checklist was added at the end of surgery, confirming the VTE risk assessment was completed and appropriate prophylaxis prescribed.
Results of compliance for all patients in each specialty were fed back weekly to all surgical specialties (sample size being 70-150 patients per week). The results were also presented at Surgical Governance meetings, adding an element of competition, which supported improvement.
These interventions led to 90% of elective surgical patients having a VTE risk assessment on admission.
The learning was that each intervention implemented had engaged all of the staff involved and importantly led to an increase in confidence and ownership of the process by the surgeons involved. The process was then rolled out to all other specialties and although there was some resistance from a few surgeons, this was counteracted by the consultants already engaged in the process and this peer pressure proved successful. Facilitation was decreased over two weeks and improvements were sustained following removal of the facilitation.
PDSA 3 was then commenced with the aim of achieving 100% compliance with completion of the VTE risk assessment.
The final intervention was the introduction of an innovative ‘clot stop’, where patients could only leave for theatre from the Admission Suite if the VTE risk assessment was completed. This was introduced with the support of the Surgical Clinical Governance Committee and meant surgeons would potentially be called back from theatre to complete the VTE risk assessment, possibly resulting in theatre delays. The implications were therefore significant, but we hypothesised that following the previous improvement, this would not prove to be too disruptive. It was important only to introduce this once we had engaged the majority of the surgeons and already had a reasonably reliable process with 90% compliance.
The ‘clot stop’ was embraced by all staff and resulted in 100% compliance with the VTE risk assessment completed on admission.
Alongside this work the Safer Clinical Systems VTE project team reviewed VTE mechanical devices in the Trust and identified three different types of mechanical devices in use. A full evaluation was undertaken and various types of devices were assessed by all stakeholders resulting in the procurement of one device. This standardisation resulted in a cost saving of £52,000. The education programme resulted in an increase in use of mechanical devices across the Trust. Other interventions to increase awareness included the re-design of the webpage for all staff, and adding the VTE guidelines to the existing hospital ‘junior doctor survival’ App. Patients were also included in the awareness campaign through a safety message created and broadcast by staff on the hospital radio, and information published in the local hospital magazine.⇓
Percentage elective patients with VTE risk assessment completed in the Admission Suite without call back: baseline March 2015 to project end - September 2015
Results
Baseline data was collected with compliance of VTE risk assessment recording on the drug chart prior to the implementation of the new system in the Admission Suite and this continued throughout each PDSA intervention. These results were then displayed in a simple run chart and the Surgical Division Clinical Lead fed back weekly to the surgical specialties.
Following introduction of the ‘clot stop’ we achieved 100% of patients leaving the Admission Suite with a completed VTE risk assessment, with about 10% of surgeons re-called from theatre. Prior to implementation of the ‘clot stop’, there had been concern that an unintended consequence would result in a delay in theatre operating start times if a surgeon was recalled to complete the VTE risk assessment. However there were no unexpected consequences reported during implementation of the new system, and none reported since the project end in September 2015.
This improvement in VTE processes has resulted in an increase in patient safety and resulted in an 80% reduction in hospital associated thrombosis following surgery from 0.2% in January 2014 to 0.04% in December 2015.
A reduction in the number of all hospital associated VTE also occurred from a baseline median of 9 per month in January 2014 to a median of 1 per month by December 2015.
Lessons and limitations
The main challenge we discovered at the start of the project was the lack of ownership by individual teams or clinicians, and the need to convey the fact that the prevention of VTE was ‘everyone's concern’. Once clarity of responsibility was agreed and communicated, the culture began to change. A robust communication strategy was developed at the beginning of the project with the Trust communications team, and information was delivered regarding the progress of the project throughout the year.
There were few limitations with this project. It was clear that there was a strong willingness from the clinicians to improve the Trust VTE performance, involving teams from the beginning and communicating progress throughout resulted in good ‘buy in’.
Engagement of senior clinical staff to motivate and lead the clinical teams was crucial, as was the leadership of the surgical division lead to provide effective and authoritative communication to surgical teams.
Implementing the new process using the “Safer Clinical Systems” quality improvement approach with the Model for Improvement was essential to achieving success. Testing the process rather than enforcing it without understanding constraints or time pressures on surgeons was important and learning from the testing was paramount. The project team took time to pause following initial testing and discuss ways in which to get engagement from the surgical teams. This led to the idea of placing the project manager in the Admission Suite for a few weeks to facilitate and help surgeons to easily ask questions, clarify the process, troubleshoot and gain education in a non-threatening interaction. They felt supported in the process and then took ownership of it.
The introduction of this new system for elective surgical patients was cost neutral and is now embedded and considered normal practice. The Admission Suite staff are empowered to activate a ‘clot stop’ if a VTE risk assessment has not been completed, but this occurs rarely. The extra check at the Sign Out of the WHO Surgical Safety Checklist further reinforces that this remains sustained.
The improvement in VTE risk assessment has continued and the process is embedded as routine practice. Data for compliance is now recorded electronically and remains over 90%. Work is being established to ensure regular feedback of electronic data for compliance monthly to ensure the improvement is sustained. The incidence of Hospital Associated Thrombosis is reported on a monthly basis.
Conclusion
The use of the Safer Clinical Systems approach7 allowed the examination of a system through a structured, methodical process. The ability to have time and personnel to focus on the risks in a system and explore the causes with a systematic approach is not common within the pressurized environments of health care, but without this in-depth knowledge and the full involvement of the staff working within the system, a sustainable re-design of a new system within the elective surgical pathway would not have been possible. Once the system had been redesigned using the Safer Clinical Systems approach, it was then essential to test the process using the Model for Improvement and learn from the testing before expanding more widely. The time spent understanding how the process was working with the teams involved resulted in ownership of the process, and was a key factor in achieving a sustainable change. This has resulted in an 80% reduction in hospital associated thrombosis from 0.2% in January 2014 to 0.04% in December 2015 of all patients undergoing a surgical procedure.
Acknowledgments
The Health Foundation who provided funding for this project
Leicester University - project evaluation
Warwick Medical School, Safer Clinical Systems Technical Support Team
Sue Scott, Senior Thrombosis Specialist Nurse, Safer Clinical Systems Project Nurse
Rachel Phillips, Thrombosis Specialist Nurse
Footnotes
Declaration of interests Professor Carol Peden has been funded by the Health Foundation as a Quality Improvement Fellow.
Ethical approval This project was exempt from ethical approval as the work was deemed an improvement study and not a study on human subjects. Local policy stated that ethical approval was not required.